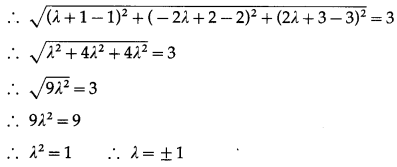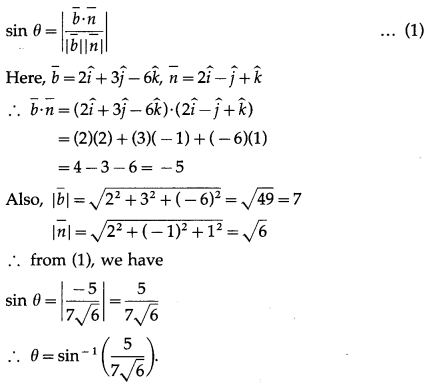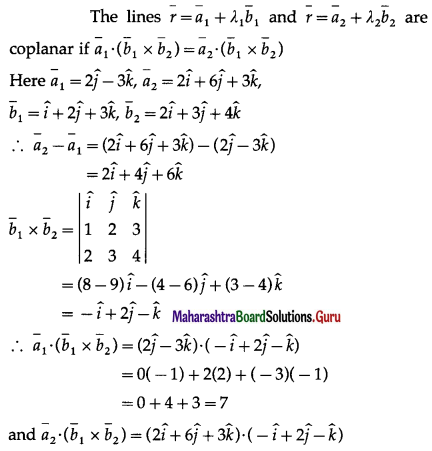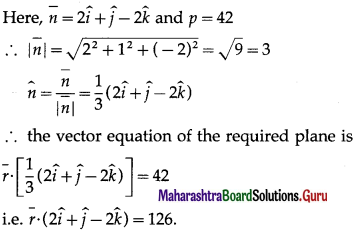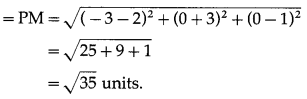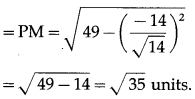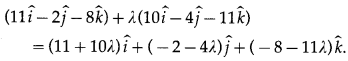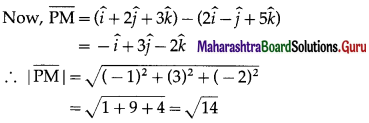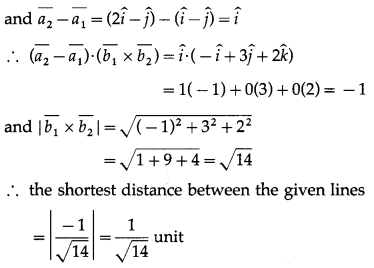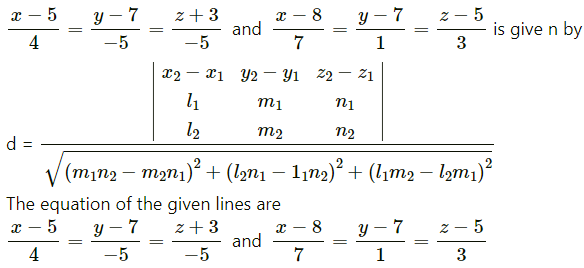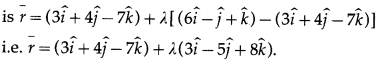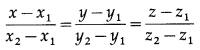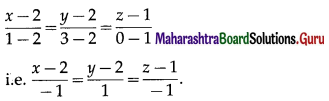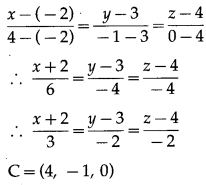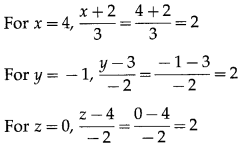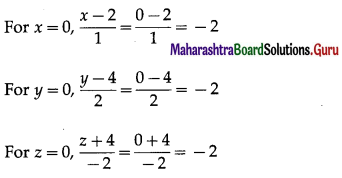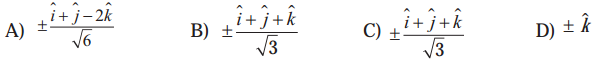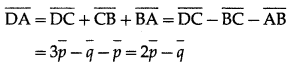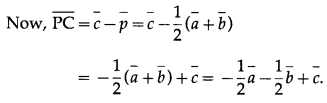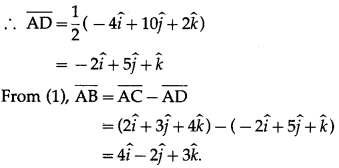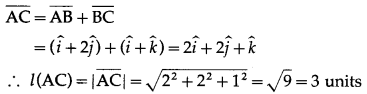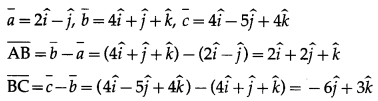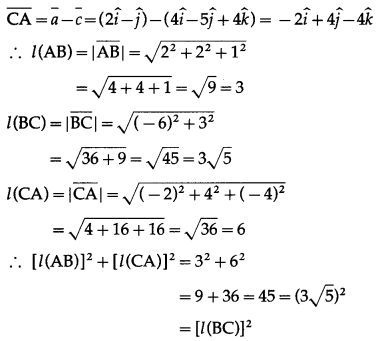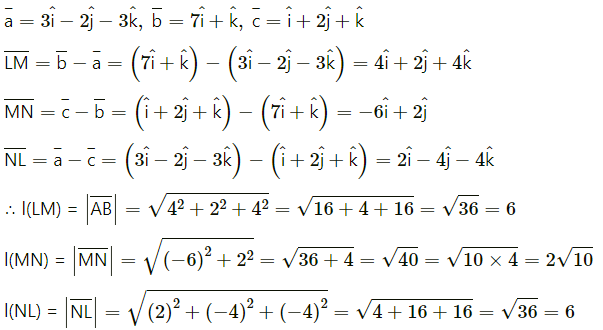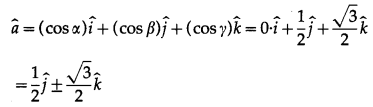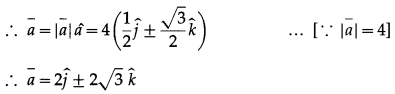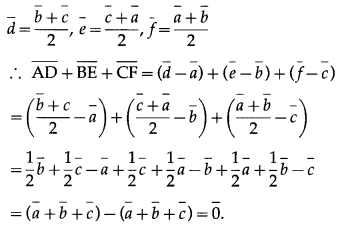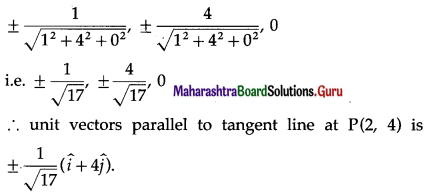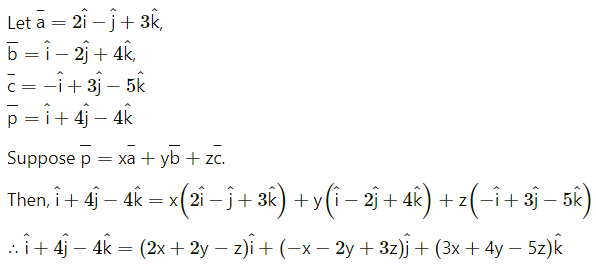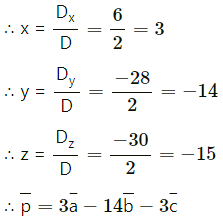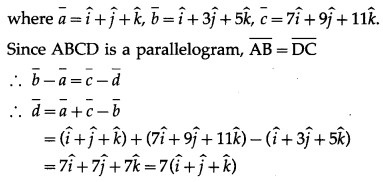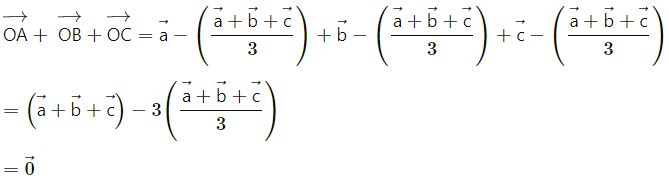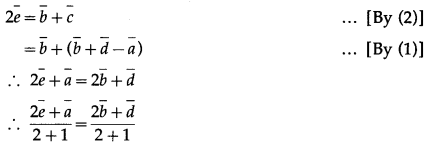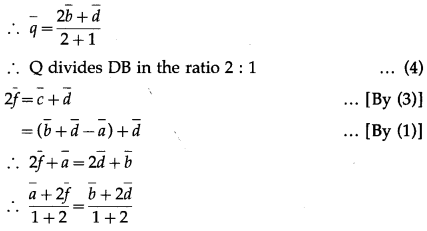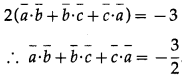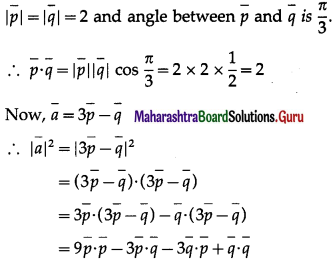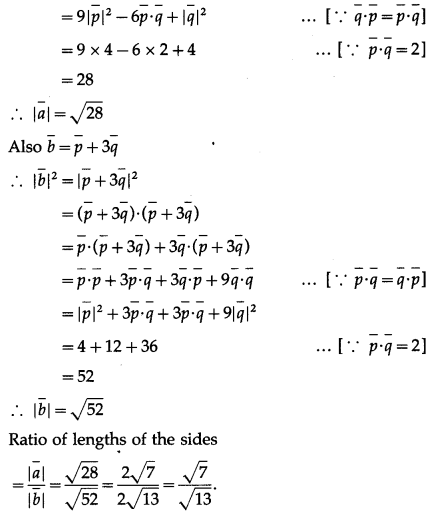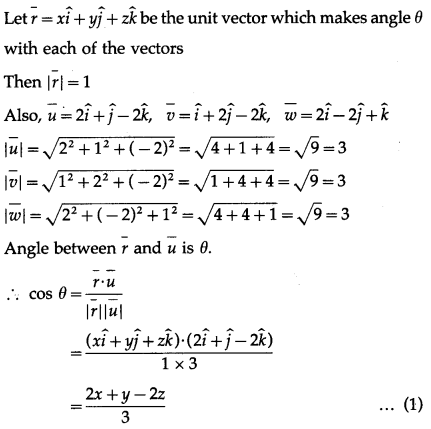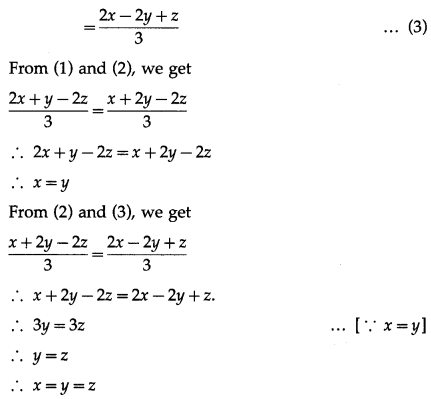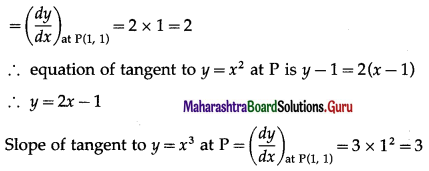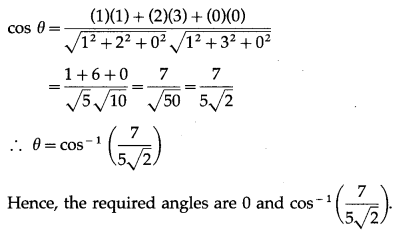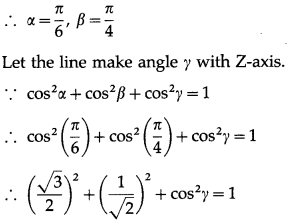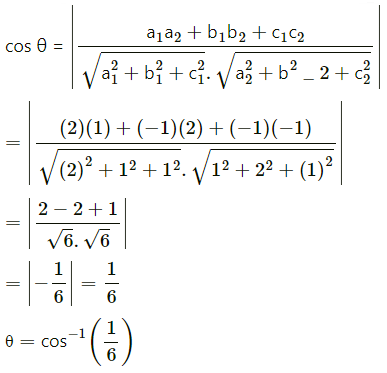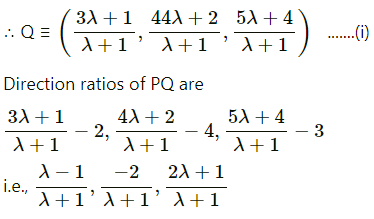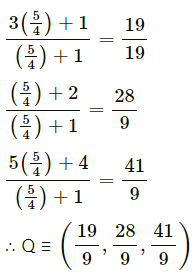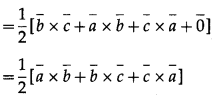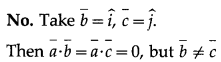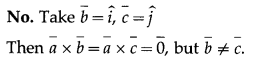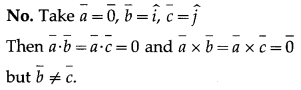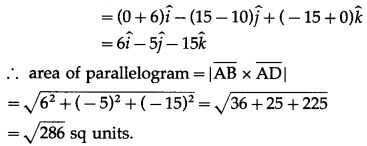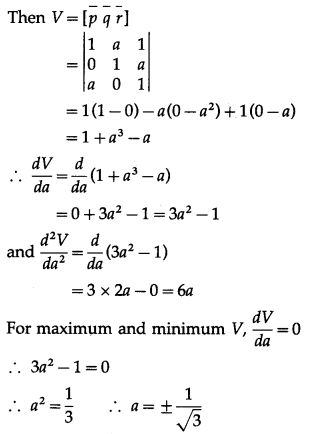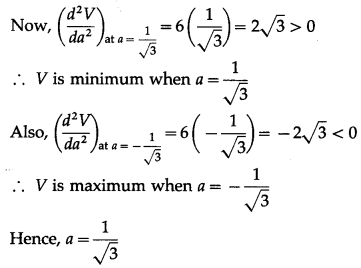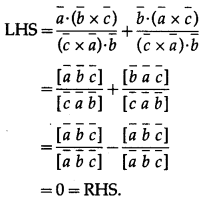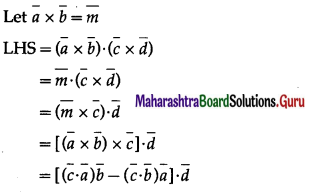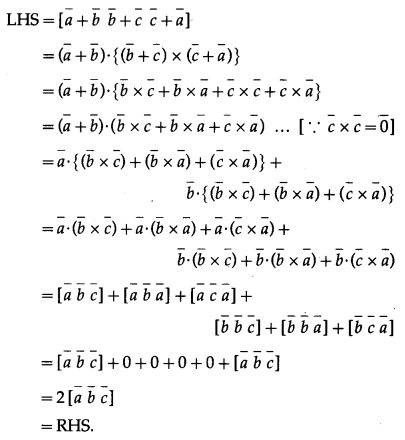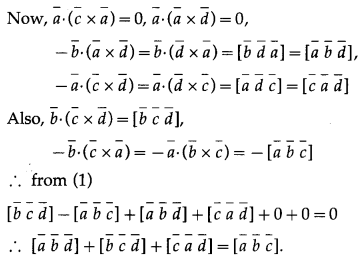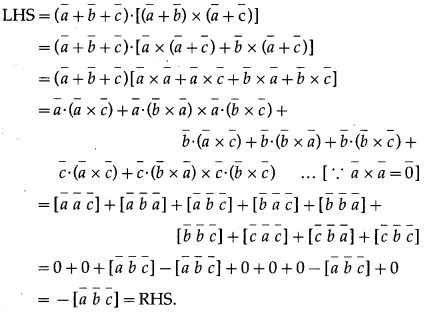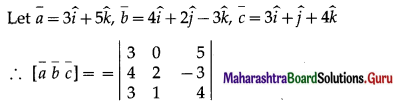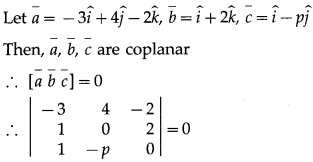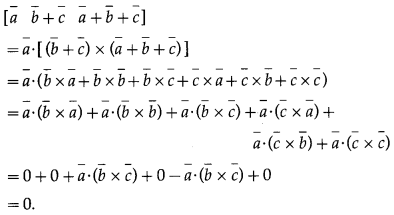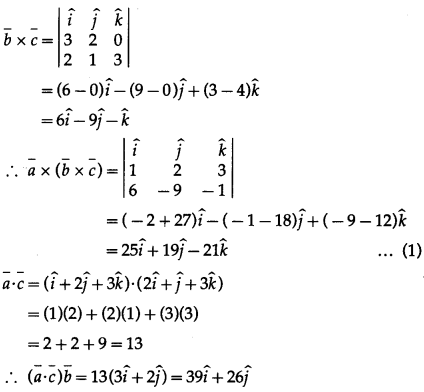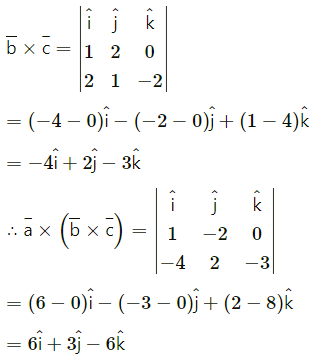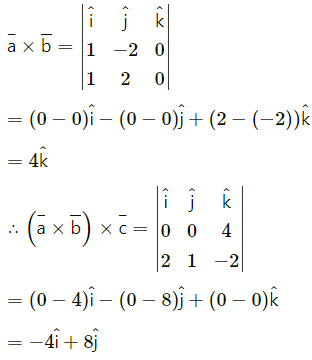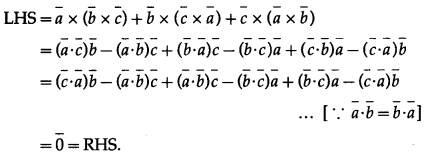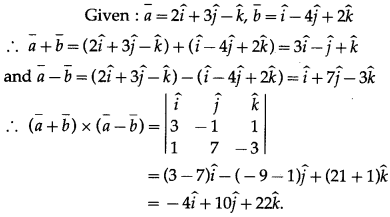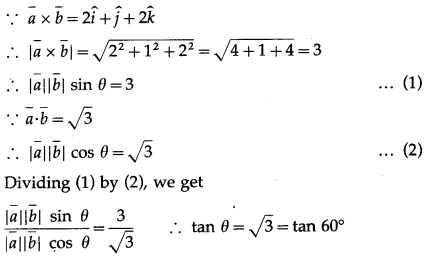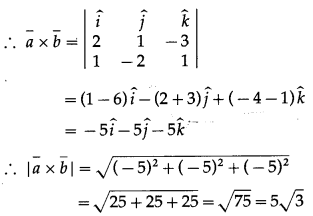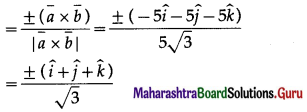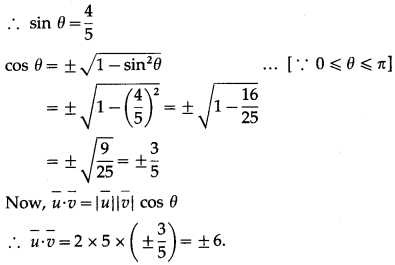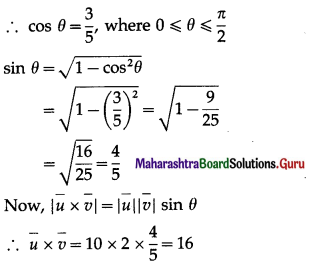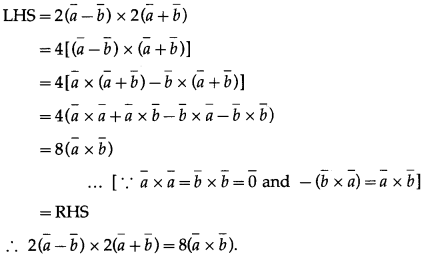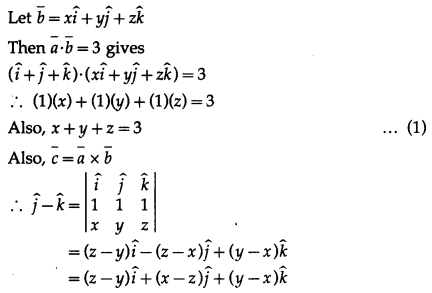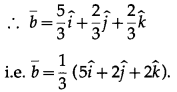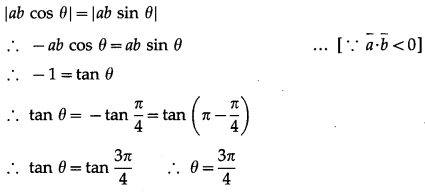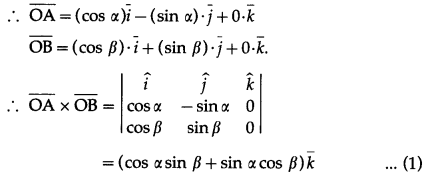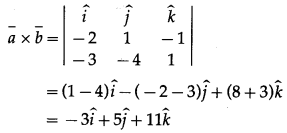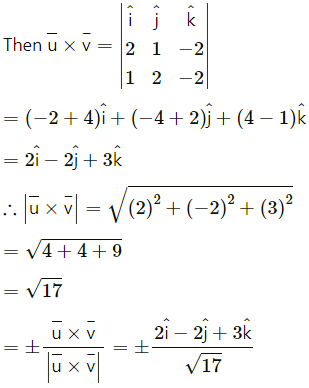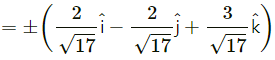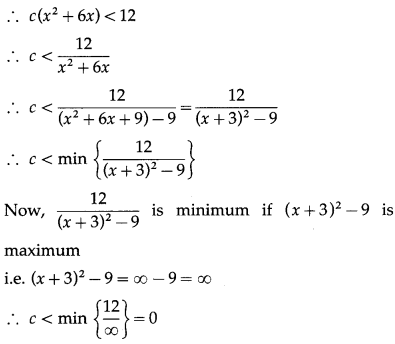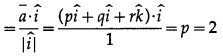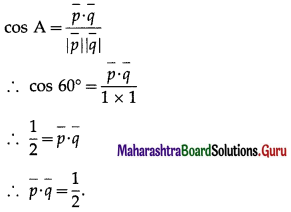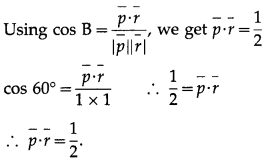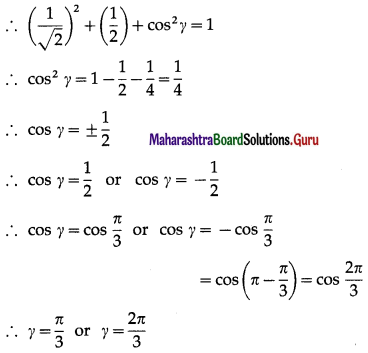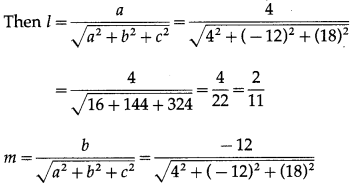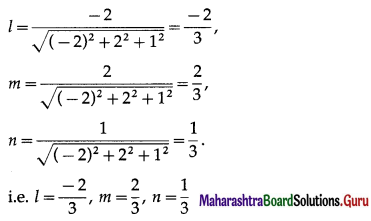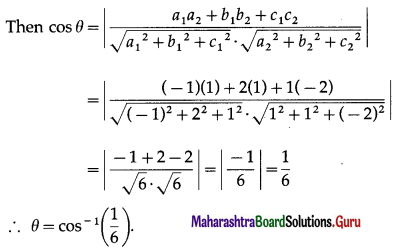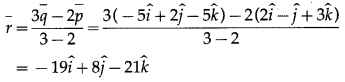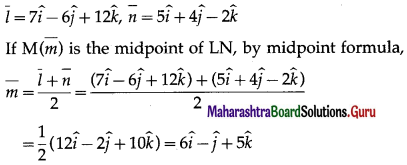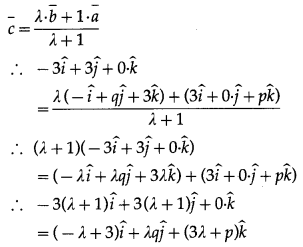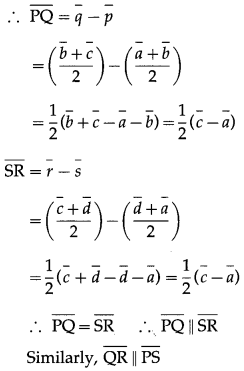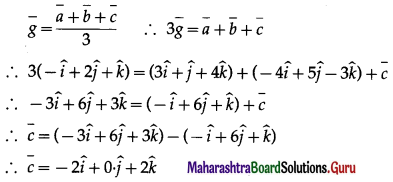Balbharti 12th Maharashtra State Board Maths Solutions Book Pdf Chapter 5 Vectors Miscellaneous Exercise 5 Questions and Answers.
12th Maths Part 1 Vectors Miscellaneous Exercise 5 Questions And Answers Maharashtra Board
I) Select the correct option from the given alternatives :
Question 1.
If |\(\bar{a}\)| = 2, |\(\bar{b}\)| = 3 |\(\bar{c}\)| = 4 then [\(\bar{a}\) + \(\bar{b}\) \(\bar{b}\) + \(\bar{c}\) \(\bar{c}\) – \(\bar{a}\)] is equal to
(A) 24
(B) -24
(C) 0
(D) 48
Solution:
(C) 0
Question 2.
If |\(\bar{a}\)| = 3, |\(\bar{b}\)| = 4, then the value of λ for which \(\bar{a}\) + λ\(\bar{b}\) is perpendicular to \(\bar{a}\) – λ\(\bar{b}\), is

Solution:
(b) \(\frac{3}{4}\)
Question 3.
If sum of two unit vectors is itself a unit vector, then the magnitude of their difference is
(A) \(\sqrt {2}\)
(B) \(\sqrt {3}\)
(C) 1
(D) 2
Solution:
(B) \(\sqrt {3}\)

Question 4.
If |\(\bar{a}\)| = 3, |\(\bar{b}\)| = 5, |\(\bar{c}\)| = 7 and \(\bar{a}\) + \(\bar{b}\) + \(\bar{c}\) = 0, then the angle between \(\bar{a}\) and \(\bar{b}\) is

Solution:
(b) \(\frac{\pi}{3}\)
Question 5.
The volume of tetrahedron whose vertices are (1, -6, 10), (-1, -3, 7), (5, -1, λ) and (7, -4, 7) is 11 cu. units then the value of λ is
(A) 7
(B) \(\frac{\pi}{3}\)
(C) 1
(D) 5
Solution:
(A) 7
Question 6.
If α, β, γ are direction angles of a line and α = 60º, β = 45º, the γ =
(A) 30º or 90º
(B) 45º or 60º
(C) 90º or 30º
(D) 60º or 120º
Solution:
(D) 60º or 120º
Question 7.
The distance of the point (3, 4, 5) from Y- axis is
(A) 3
(B) 5
(C) \(\sqrt {34}\)
(D) \(\sqrt {41}\)
Solution:
(C) \(\sqrt {34}\)

Question 8.
The line joining the points (-2, 1, -8) and (a, b, c) is parallel to the line whose direction ratios are 6, 2, 3. The value of a, b, c are
(A) 4, 3, -5
(B) 1, 2, \(\frac{-13}{2}\)
(C) 10, 5, -2
(D) 3, 5, 11
Solution:
(A) 4, 3, -5
Question 9.
If cos α, cos β, cos γ are the direction cosines of a line then the value of sin2 α + sin2β + sin2γ is
(A) 1
(B) 2
(C) 3
(D) 4
Solution:
(B) 2
Question 10.
If l, m, n are direction cosines of a line then \(\hat{l}+m \hat{j}+n \hat{k}\) is
(A) null vector
(B) the unit vector along the line
(C) any vector along the line
(D) a vector perpendicular to the line
Solution:
(B) the unit vector along the line
Question 11.
If |\(\bar{a}\)| = 3 and –1 ≤ k ≤ 2, then |k\(\bar{a}\)| lies in the interval
(A) [0, 6]
(B) [-3, 6]
(C) [3, 6]
(D) [1, 2]
Solution:
(A) [0, 6]
Question 12.
Let α, β, γ be distinct real numbers. The points with position vectors \(\alpha \hat{i}+\beta \hat{j}+\gamma \hat{k}\), \(\beta \hat{i}+\gamma \hat{j}+\alpha \hat{k}\), \(\gamma \hat{i}+\alpha \hat{j}+\beta \hat{k}\)
(A) are collinear
(B) form an equilateral triangle
(C) form a scalene triangle
(D) form a right angled triangle
Solution:
(B) form an equilateral triangle

Question 13.
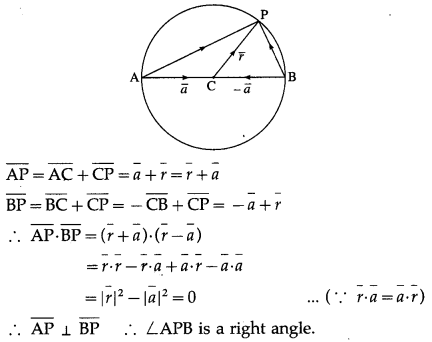
Let \(\bar{p}\) and \(\bar{q}\) be the position vectors of P and Q respectively, with respect to O and |\(\bar{p}\)| = p, |\(\bar{q}\)| = q. The points R and S divide PQ internally and externally in the ratio 2 : 3 respectively. If OR and OS are perpendicular then.
(A) 9p2 = 4q2
(B) 4p2 = 9q2
(C) 9p = 4q
(D) 4p = 9q
Solution:
(A) 9p2 = 4q2
Question 14.
The 2 vectors \(\hat{j}+\hat{k}\) and \(3 \hat{i}-\hat{j}+4 \hat{k}\) represents the two sides AB and AC, respectively of a ∆ABC. The length of the median through A is
(A) \(\frac{\sqrt{34}}{2}\)
(B) \(\frac{\sqrt{48}}{2}\)
(C) \(\sqrt {18}\)
(D) None of these
Solution:
(A) \(\frac{\sqrt{34}}{2}\)
Question 15.
If \(\bar{a}\) and \(\bar{b}\) are unit vectors, then what is the angle between \(\bar{a}\) and \(\bar{b}\) for \(\sqrt{3} \bar{a}\) – \(\bar{b}\) to be a unit vector ?
(A) 30º
(B) 45º
(C) 60º
(D) 90º
Solution:
(A) 30º
Question 16.
If θ be the angle between any two vectors \(\bar{a}\) and \(\bar{b}\), then \(|\vec{a} \cdot \vec{b}|\) = \(|\vec{a} \times \vec{b}|\), when θ is equal to
(A) 0
(B) \(\frac{\pi}{4}\)
(C) \(\frac{\pi}{2}\)
(D) π
Solution:
(B) \(\frac{\pi}{4}\)

Question 17.
The value of \(\hat{i} \cdot(\hat{j} \times \hat{k})+\hat{j} \cdot(\hat{i} \times \hat{k})+\hat{k} \cdot(\hat{i} \times \hat{j})\)
(A) 0
(B) -1
(C) 1
(D) 3
Solution:
(C) 1
Question 18.
Let a, b, c be distinct non-negative numbers. If the vectors \(\mathrm{a} \hat{i}+\mathrm{a} \hat{j}+\mathrm{c} \hat{k}\), \(\hat{i}+\hat{k}\) and \(\mathbf{c} \hat{i}+\mathrm{c} \hat{j}+\mathrm{b} \hat{k}\) lie in a plane, then c is
(A) The arithmetic mean of a and b
(B) The geometric mean of a and b
(C) The harmonic man of a and b
(D) 0
Solution:
(B) The geometric mean of a and b
Question 19.
Let \(\bar{a}\) = \(\hat{i} \hat{j}\), \(\bar{b}\) = \(\hat{j} \hat{k}\), \(\bar{c}\) = \(\hat{k} \hat{i}\). If \(\bar{d}\) is a unit vector such that \(\bar{a} . \bar{d}=0=\left[\begin{array}{lll}
\bar{b} & \bar{c} & \bar{d}
\end{array}\right]\), then \(\bar{d}\) equals.
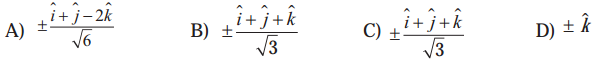
Solution:
(a) \(\pm \frac{\hat{i}+\hat{j}-2 \hat{k}}{\sqrt{6}}\)
Question 20.
If \(\bar{a}\) \(\bar{b}\) \(\bar{c}\) are non coplanar unit vectors such that \(\bar{a} \times(\bar{b} \times \bar{c})\) =\(\frac{(\bar{b}+\bar{c})}{\sqrt{2}}\) then the angle between \(\bar{a}\) and \(\bar{b}\) is
(A) \(\frac{3 \pi}{4}\)
(B) \(\frac{\pi}{4}\)
(C) \(\frac{\pi}{2}\)
(D) π
Solution:
(A) \(\frac{3 \pi}{4}\)

II Answer the following :
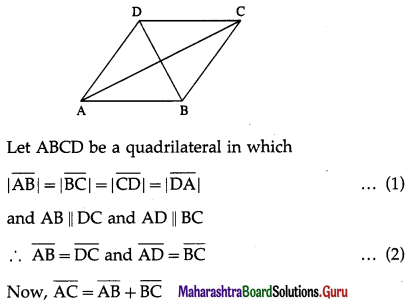
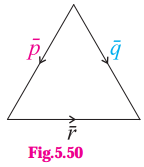
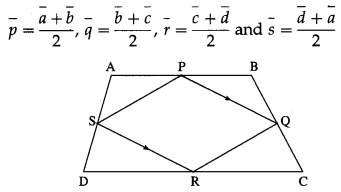
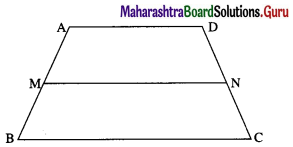
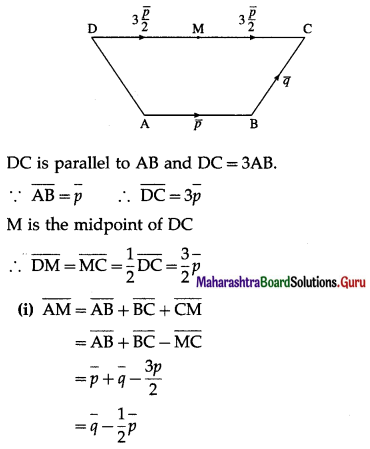
1) ABCD is a trapezium with AB parallel to DC and DC = 3AB. M is the mid-point of DC,
\(\overline{A B}\) = \(\bar{p}\) and \(\overline{B C}\) = \(\bar{q}\). Find in terms of \(\bar{p}\) and \(\bar{q}\).
(i) \(\overline{A M}\)
Solution:
(ii) \(\overline{B D}\)
Solution:
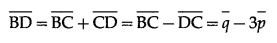
(iii) \(\overline{M B}\)
Solution:
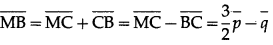
(iv) \(\overline{D A}\)
Solution:
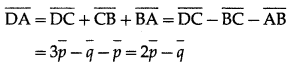
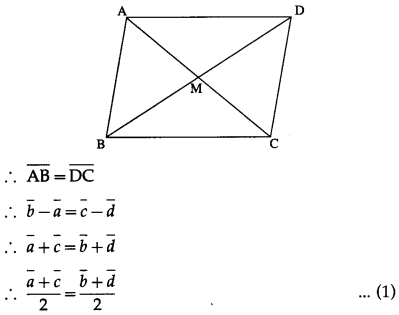
Question 2.
The points A, B and C have position vectors \(\bar{a}\), \(\bar{b}\) and \(\bar{c}\) respectively. The point P is midpoint of AB. Find in terms of \(\bar{a}\), \(\bar{b}\) and \(\bar{c}\) the vector \(\overline{P C}\)
Solution:
P is the mid-point of AB.
∴ \(\bar{p}\) = \(=\frac{\bar{a}+\bar{b}}{2}\), where \(\bar{p}\) is the position vector of P.
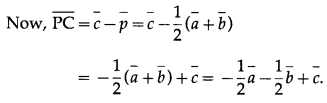
Question 3.
In a pentagon ABCDE
Show that \(\overline{A B}\) + \(\overline{A E}\) + \(\overline{B C}\) + \(\overline{D C}\) + \(\overline{E D}\) = 2\(\overline{A C}\)
Solution:
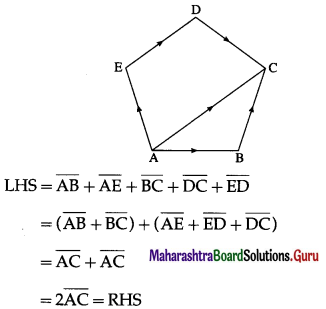
Question 4.
If in parallelogram ABCD, diagonal vectors are \(\overline{A C}\) = \(2 \hat{i}+3 \hat{j}+4 \hat{k}\) and \(\overline{B D}\) = \(-6 \hat{i}+7 \hat{j}-2 \hat{k}\), then find the adjacent side vectors \(\overline{A B}\) and \(\overline{A D}\)
Solution:
ABCD is a parallelogram
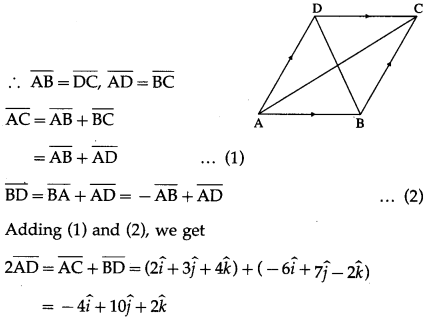
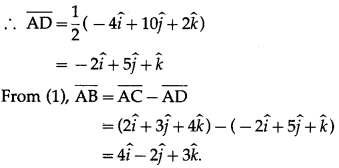

Question 5.
If two sides of a triangle are \(\hat{i}+2 \hat{j}\) and \(\hat{i}+\hat{k}\), then find the length of the third side.
Solution:
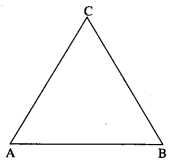
Let ABC be a triangle with \(\overline{A B}\) = \(\hat{i}+2 \hat{j}\), \(\overline{B C}\) = \(\hat{i}+\hat{k}\).
By triangle law of vectors
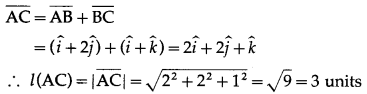
Hence, the length of third side is 3 units.
Question 6.
If |\(\bar{a}\)| = |\(\bar{b}\) | = 1 \(\bar{a}\).\(\bar{b}\) = 0 and \(\bar{a}\) + \(\bar{b}\) + \(\bar{c}\) = 0 then find |\(\bar{c}\)|
Solution:
\(\bar{a}\) + \(\bar{b}\) + \(\bar{c}\) = 0
∴ –\(\bar{c}\) = \(\bar{a}\) + \(\bar{b}\)
Taking dot product of both sides with itself, we get

Question 7.
Find the lengths of the sides of the triangle and also determine the type of a triangle.
(i) A(2, -1, 0), B(4, 1, 1,), C(4, -5, 4)
Solution:
The position vectors \(\bar{a}\), \(\bar{b}\), \(\bar{c}\) of the points A, B, C are
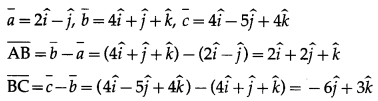
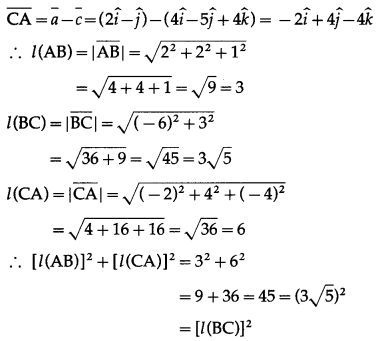
∴ ∆ ABC is right angled at A.
(ii) L(3, -2, -3), M(7, 0, 1), N (1, 2, 1)
Solution:
The position vectors bar \(\bar{a}\), \(\bar{b}\), \(\bar{c}\) of the points L M, N are
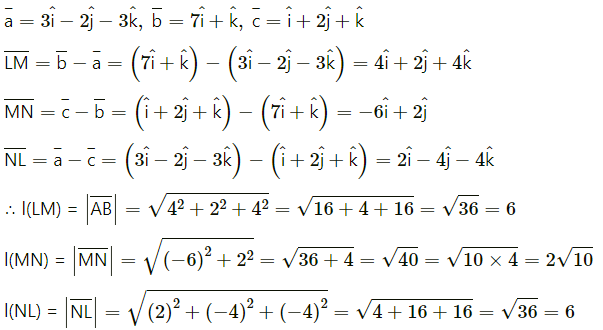
l(LM) = 6, l(MN) = 2\(\sqrt {10}\) , l(NL) = 6
∆LMN is sosceles

Question 8.
Find the component form of if a if
(i) It lies in YZ plane and makes 60º with positive Y-axis and |\(\bar{a}\)| = 4
Solution:
Let α, β, γ be the direction angles of \(\bar{a}\)
Since \(\bar{a}\) lies in YZ-plane, it is perpendicular to X-axis
∴ α = 90°
It is given that β= 60°
∵ cos2α + cos2β + cos2γ = 1
∴ cos290° + cos260° + cos2γ = 1
∴ 0 + \(\left(\frac{1}{2}\right)^{2}\) + cos2γ = 1
∴ cos2γ = 1 – \(\frac{1}{4}=\frac{3}{4}\)
∴ cos γ = \(\pm \frac{\sqrt{3}}{2}\)
Unit vector along a is given by
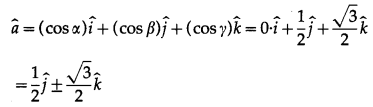
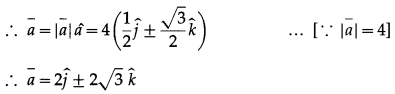
(ii) It lies in XZ plane and makes 45º with positive Z-axis and |\(\bar{a}\)| = 10
Solution:
Question 9.
Two sides of a parallelogram are \(3 \hat{i}+4 \hat{j}-5 \hat{k}\) and \(-2 \hat{j}+7 \hat{k}\). Find the unit vectors parallel to the diagonals.
Solution:
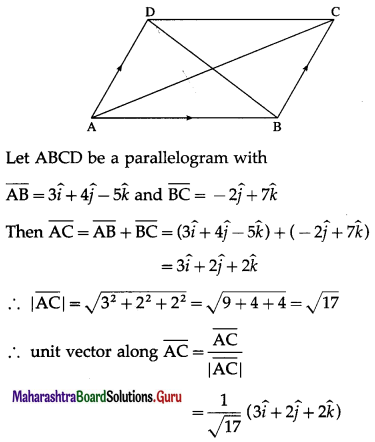

Question 10.
If D, E, F are the mid-points of the sides BC, CA, AB of a triangle ABC , prove that \(\overline{A D}\) + \(\overline{B E}\) + \(\overline{C F}\) = 0
Solution:
Let \(\bar{a}\), \(\bar{b}\), \(\bar{c}\), \(\bar{d}\), \(\bar{e}\), \(\bar{f}\) be the position vectors of the points A, B, C, D, E, F respectively.
Since D, E, F are the midpoints of BC, CA, AB respec-tively, by the midpoint formula
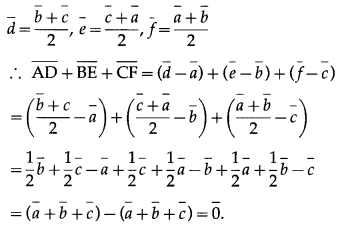
Question 11.
Find the unit vectors that are parallel to the tangent line to the parabola y = x2 at the point (2, 4)
Solution:
Differentiating y = x2 w.r.t. x, we get \(\) = 2x
Slope of tangent at P(2, 4) = \(\left(\frac{d y}{d x}\right)_{\text {at } \mathrm{P}(2, 4)}\) = 2 × 2 = 4
∴ the equation of tangent at P is
y – 4 = 4(-2)
∴ y = 4x – 4
∴ y = 4x is equation of line parallel to the tangent at P and passing through the origin O.
4x = y, z = 0 ∴ \(\frac{x}{1}=\frac{y}{4}\), z = 0
∴ the direction ratios of this line are 1, 4, 0
∴ its direction cosines are
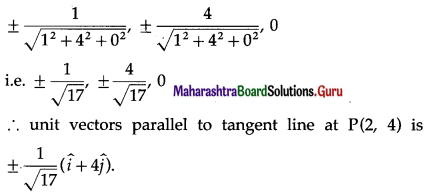

Question 12.
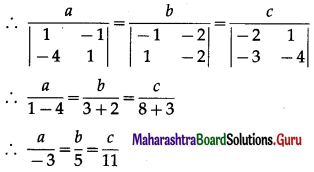
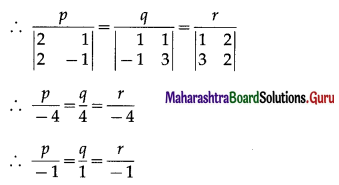
Express the vector \(\hat{i}+4 \hat{j}-4 \hat{k}\) as a linear combination of the vectors \(2 \hat{i}-\hat{j}+3 \hat{k}\), \(\hat{i}-2 \hat{j}+4 \hat{k}\) and \(-\hat{i}+3 \hat{j}-5 \hat{k}\)
Solution:
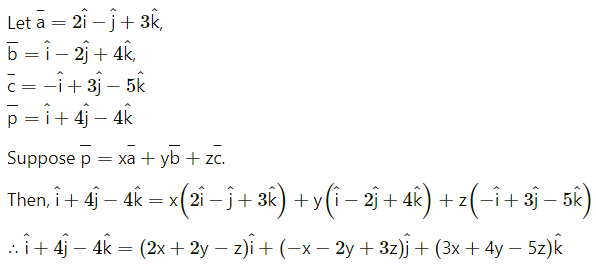
By equality of vectors,
2x + 2y – z = 1
-x – 2y + 3z = 4
3x + 4y – 5z = -4
We have to solve these equations by using Cramer’s Rule.
D = \(\left|\begin{array}{ccc}
2 & 2 & -1 \\
-1 & -2 & 3 \\
3 & 4 & -5
\end{array}\right|\)
= 2(10 – 12) – 2(5 – 9) – 1(-4 + 6)
= -4 + 8 – 2
= 2 ≠ 0
Dx = \(\left|\begin{array}{ccc}
1 & 2 & -1 \\
4 & -2 & 3 \\
-4 & 4 & -5
\end{array}\right|\)
= 1(10 – 12) – 2(-20 + 12) – 1 (16 – 8)
= -2 + 16 – 8
= 6
Dy = \(\left|\begin{array}{ccc}
2 & 1 & -1 \\
-1 & 4 & 3 \\
3 & -4 & -5
\end{array}\right|\)
= 2(-20 + 12) – 1(5 – 9) – 1(4 – 12)
= -16 – 4 – 8
= -28
Dz = \(\left|\begin{array}{ccc}
2 & 2 & 1 \\
-1 & -2 & 4 \\
3 & 4 & -4
\end{array}\right|\)
= 2(8 – 16) – 2(4 – 12) + 1(-4 + 6)
= -16 – 16 + 2
= -30
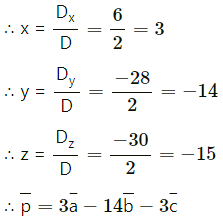
Question 13.
If \(\overline{O A}\) = \(\bar{a}\) and \(\overline{O B}\) = \(\bar{b}\) then show that the vector along the angle bisector of angle AOB is
given by \(\bar{d}\) = λ\(\left(\frac{\bar{a}}{|\bar{b}|}+\frac{\bar{b}}{|\bar{b}|}\right)\)
Question is modified
If \(\overline{O A}\) = \(\bar{a}\) and \(\overline{O B}\) = \(\bar{b}\) then show that the vector along the angle bisector of ∠AOB is
given by \(\bar{d}\) = λ\(\left(\frac{\bar{a}}{|\bar{a}|}+\frac{\bar{b}}{|\bar{b}|}\right)\)
Solution:
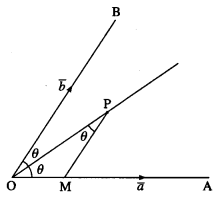
Choose any point P on the angle bisector of ∠AOB. Draw PM parallel to OB.
∴ ∠OPM = ∠POM
= ∠POB
Hence, OM = MP
∴ OM and MP is the same scalar multiple of unit vectors \(\hat{a}\) and \(\hat{b}\) along these directions,


Question 14.
The position vectors f three consecutive vertices of a parallelogram are \(\hat{i}+\hat{j}+\hat{k}\), \(\hat{i}+3 \hat{j}+5 \hat{k}\) and \(7 \hat{i}+9 \hat{j}+11 \hat{k}\) Find the position vector of the fourth vertex.
Solution:
Let ABCD be a parallelogram.
Let \(\bar{a}\), \(\bar{b}\), \(\bar{c}\), \(\bar{d}\) be the position vectors of the vertices
A, B, C, D of the parallelogram,
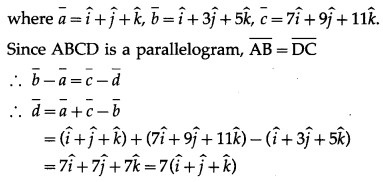
Hence, the position vector of the fourth vertex is 7(\(\hat{i}\) + \(\hat{j}\) + \(\hat{k}\)).
Question 15.
A point P with position vector \(\frac{-14 \hat{i}+39 \hat{j}+28 \hat{k}}{5}\) divides the line joining A(-1, 6, 5) and B in the ratio 3 : 2 then find the point B.
Solution:
Let A, B and P have position vectors \(\bar{a}\), \(\bar{b}\) and \(\bar{p}\) respectively.

∴ coordinates of B are (-4, 9, 6).
Question 16.
Prove that the sum of the three vectors determined by the medians of a triangle directed from the vertices is zero.
Solution:
Let \(\overrightarrow{\mathrm{a}}\), \(\overrightarrow{\mathrm{b}}\) and \(\overrightarrow{\mathrm{c}}\) are the position vectors of the vertices A, B and C respectively.
Then we know that the position vector of the centroid O of the triangle is \(\frac{\vec{a}+\vec{b}+\vec{c}}{3}\)
Therefore sum of the three vectors \(\overrightarrow{\mathrm{OA}}\), \(\overrightarrow{\mathrm{OB}}\) and \(\overrightarrow{\mathrm{OC}}\), is
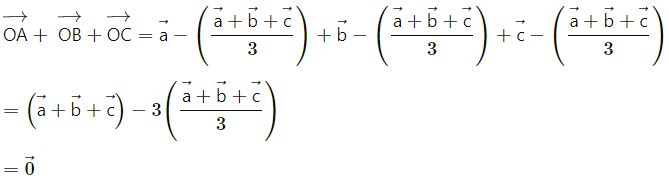
Hence, Sum os the three vectors determined by the medians of a triangle directed from the vertices is zero.
Question 17.
ABCD is a parallelogram E, F are the mid points of BC and CD respectively. AE, AF meet the diagonal BD at Q and P respectively. Show that P and Q trisect DB.
Solution:
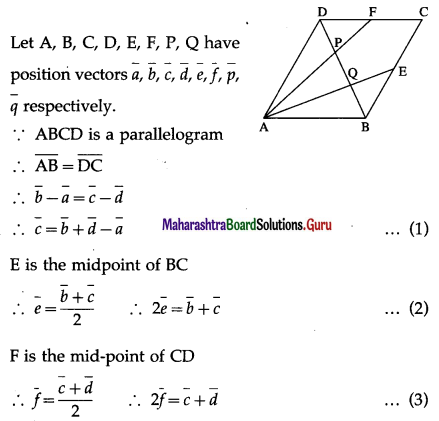
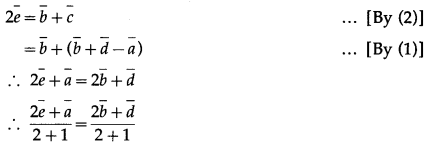
LHS is the position vector of the point on AE and RHS is the position vector of the point on DB. But AE and DB meet at Q.
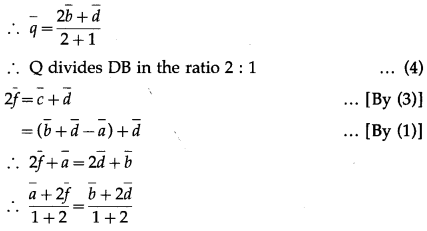
LHS is the position vector of the point on AF and RHS is the position vector of the point on DB.
But AF and DB meet at P.
∴ \(\bar{p}=\frac{\bar{b}+2 \bar{d}}{1+2}\)
∴ P divides DB in the ratio 1 : 2 … (5)
From (4) and (5), if follows that P and Q trisect DB.

Question 18.
If aBC is a triangle whose orthocenter is P and the circumcenter is Q, then prove that \(\overline{P A}\) + \(\overline{P C}\) + \(\overline{P B}\) = 2 \(\overline{P Q}\)
Solution:
Let G be the centroid of the ∆ ABC.
Let A, B, C, G, Q have position vectors \(\bar{a}\), \(\bar{b}\), \(\bar{c}\), \(\bar{g}\), \(\bar{q}\) w.r.t. P. We know that Q, G, P are collinear and G divides segment QP internally in the ratio 1 : 2.

Question 19.
If P is orthocenter, Q is circumcenter and G is centroid of a triangle ABC, then prove that \(\overline{Q P}\) = 3\(\overline{Q G}\)
Solution:
Let \(\bar{p}\) and \(\bar{g}\) be the position vectors of P and G w.r.t. the circumcentre Q.
i.e. \(\overline{\mathrm{QP}}\) = p and \(\overline{\mathrm{QG}}\) = g.
We know that Q, G, P are collinear and G divides segment QP internally in the ratio 1 : 2
∴ by section formula for internal division,

Question 20.
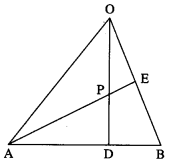
In a triangle OAB, E is the midpoint of BO and D is a point on AB such that AD: DB = 2:1. If OD and AE intersect at P, determine the ratio OP:PD using vector methods.
Solution:
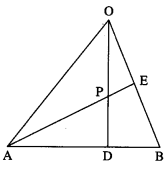
Let A, B, D, E, P have position vectors \(\bar{a}\), \(\bar{b}\), \(\bar{d}\), \(\bar{e}\), \(\bar{p}\) respectively w.r.t. O.
∵ AD : DB = 2 : 1.
∴ D divides AB internally in the ratio 2 : 1.
Using section formula for internal division, we get

LHS is the position vector of the point which divides OD internally in the ratio 3 : 2.
RHS is the position vector of the point which divides AE internally in the ratio 4 : 1.
But OD and AE intersect at P
∴ P divides OD internally in the ratio 3 : 2.
Hence, OP : PD = 3 : 2.
Question 21.
Dot-product of a vector with vectors \(3 \hat{i}-5 \hat{k}, 2 \hat{i}+7 \hat{j}\) and \(\hat{i}+\hat{j}+\hat{k}\) are respectively -1, 6 and 5. Find the vector.
Solution:

∴ 3x – 5z= -1 … (1)
∴ 2x + 7y = 6 … (2)
∴ x + y + z = 5 … (3)
From (3), z = 5 – x – y
Substituting this value of z in (1), we get
∴ 3x – 5(5 – x – y)= -1
∴ 8x + 5y = 24 … (4)
Multiplying (2) by 4 and subtracting from (4), we get
8x + 5y – 4(2x + 7y) = 24 – 6 × 4
∴ -23y = 0 ∴ y = 0
Substituting y = 0 in (2), we get
∴ 2x = 6 ∴ x = 3
Substituting x = 3 in (1), we get
∴ 3(3) – 5z = -1
∴ 5z = -10 ∴ z = 2
∴ \(\bar{r}=3 \hat{i}+0 \cdot \hat{j}+2 \hat{k}=3 \hat{i}+2 \hat{k}\)
Hence, the required vector is \(3 \hat{i}+2 \hat{k}\)

Question 22.
If \(\bar{a}\), \(\bar{b}\), \(\bar{c}\) are unit vectors such that \(\bar{a}\) + \(\bar{b}\) + \(\bar{c}\) = 0, then find the value of \(\bar{a}\).\(\bar{b}\) + \(\bar{b}\).\(\bar{c}\) + \(\bar{c}\).\(\bar{a}\)
Solution:
\(\bar{a}\), \(\bar{b}\), \(\bar{c}\) are unit vectors

Adding (2), (3), (4) and using the fact that scalar product commutative, we get
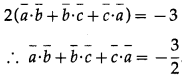
Question 23.
If a parallelogram is constructed on the vectors \(\bar{a}=3 \bar{p}-\bar{q}\), \(\bar{b}=\bar{p}+3 \bar{q}\) and \(|\bar{p}|=|\bar{q}|=2\) and angle between \(\bar{p}\) and \(\bar{q}\) is\(\frac{\pi}{3}\) show that the ratio of the lengths of the sides is \(\sqrt {7}\) : \(\sqrt {13}\)
Solution:
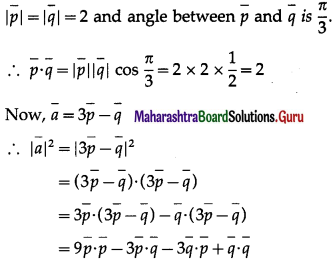
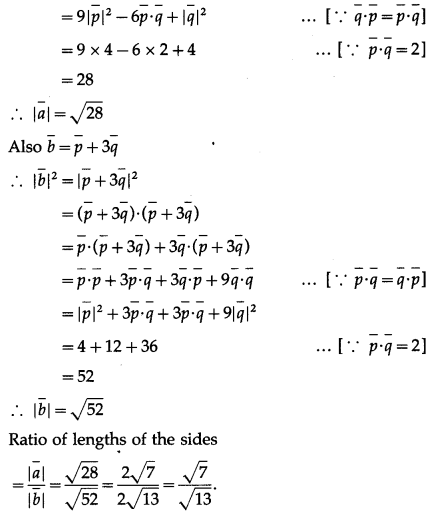
Hence, the ratio of the lengths of the sides is \(\sqrt {7}\) : \(\sqrt {13}\).
Question 24.
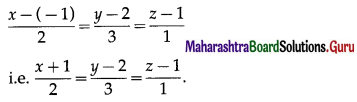
Express the vector \(\bar{a}=5 \hat{i}-2 \hat{j}+5 \hat{k}\) as a sum of two vectors such that one is parallel to the vector \(\bar{b}=3 \hat{i}+\hat{k}\) and other is perpendicular to \(\bar{b}\).
Solution:

By equality of vectors
3m + x = 5 … (1)
y = -2
and m – 3x = 5
From (1) and (2)
3m + x = m – 3x
∴ 2m = -4x m ∴ m = -2x
Substituting m = -2x in (1), we get
∴ -6x + x = 5 ∴ -5x = 5 ∴ x = -1
∴ m = -2x = 2


Question 25.
Find two unit vectors each of which makes equal angles with \(\bar{u}\), \(\bar{v}\) and \(\bar{w}\). \(\bar{u}=2 \hat{i}+\hat{j}-2 \hat{k}\), \(\bar{v}=\hat{i}+2 \hat{j}-2 \hat{k}\) and \(\bar{W}=2 \hat{i}-2 \hat{j}+\hat{k}\)
Solution:
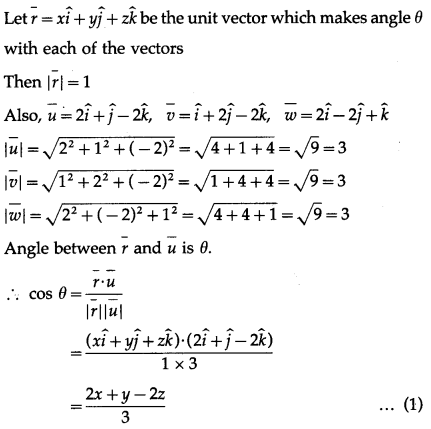

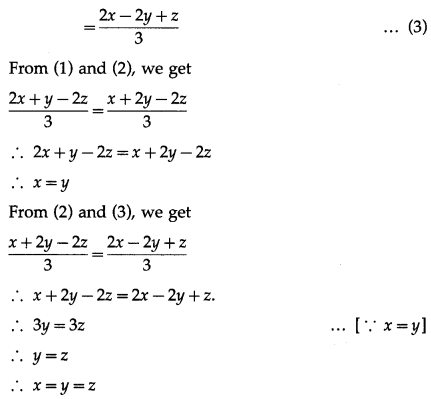

Question 26.
Find the acute angles between the curves at their points of intersection. y = x2, y = x3
Solution:
The angle between the curves is same as the angle between their tangents at the points of intersection. We find the points of intersection of y = x2 … (1)
and y = x3 … (2)
From (1) and (2)
x3 = x2
∴ x3 – x2 = 0
∴ x2(x – 1) = 0
∴ x = 0 or x = 1
When x = 0, y = 0.
When x = 1, y = 1.

∴ equation of tangent to y = x3 at P is y = 0.
∴ the tangents to both curves at (0, 0) are y = 0
∴ angle between them is 0.
Angle at P = (1, 1)
Slope of tangent to y = x2 at P
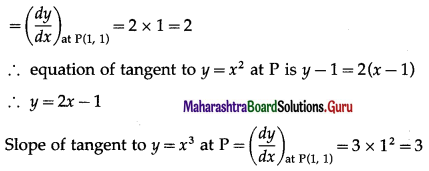
∴ equation of tangent to y = x3 at P is y – 1 = 3(x – 1) y = 3x – 2
We have to find angle between y = 2x – 1 and y = 3x – 2
Lines through origin parallel to these tagents are y = 2x and y = 3x
∴ \(\frac{x}{1}=\frac{y}{2}\) and \(\frac{x}{1}=\frac{y}{3}\)
These lines lie in XY-plane.
∴ the direction ratios of these lines are 1, 2, 0 and 1, 3, 0.
The angle θ between them is given by
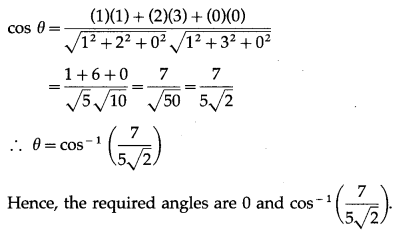
Question 27.
Find the direction cosines and direction angles of the vector.
(i) \(2 \hat{i}+\hat{j}+2 \hat{k}\)
Solution:
Let \(\bar{a}\) = \(2 \hat{i}+\hat{j}+2 \hat{k}\)

(ii) \((1 / 2) \hat{i}+\hat{j}+\hat{k}\)
Solution:

Question 28.
Let \(\bar{b}\) = \(4 \hat{i}+3 \hat{j}\) and \(\bar{c}\) be two vectors perpendicular to each other in the XY-plane. Find vectors in the same plane having projection 1 and 2 along \(\bar{b}\) and \(\bar{c}\), respectively, are given y.
Solution:
\(\bar{b}\) = \(4 \hat{i}+3 \hat{j}\)



Question 29.
Show that no line in space can make angle \(\frac{\pi}{6}\) and \(\frac{\pi}{4}\) with X- axis and Y-axis.
Solution:
Let, if possible, a line in space make angles \(\frac{\pi}{6}\) and \(\frac{\pi}{4}\) with X-axis and Y-axis.
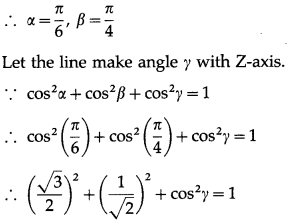
∴ cos2γ = 1 – \(\frac{3}{4}-\frac{1}{2}=-\frac{1}{4}\)
This is not possible, because cos γ is real
∴ cos2γ cannot be negative.
Hence, there is no line in space which makes angles \(\frac{\pi}{6}\) and \(\frac{\pi}{4}\) with X-axis and Y-axis.
Question 30.
Find the angle between the lines whose direction cosines are given by the equation 6mn – 2nl + 5lm = 0, 3l + m + 5n = 0
Solution:
Given 6mn – 2nl + 5lm = o
3l + m +5n = 0.
From (2), m = 3l – 5n
Putting the value of m in equation (1), we get,
⇒ 6n(-3l – 5n) – 2nl + 5l(-3l – 5n) = 0
⇒ -18nl- 30n – 2nl- 15l2 – 25nl = 0
⇒ – 30n2 – 45nl – 15l2 = 0
⇒ 2n2 + 3nl + l2 = 0
⇒ 2n2 + 2nl + nl + l2 = 0
⇒ (2n + l) (n + l) = 0
∴ 2n + l = 0 OR n + l = 0
∴ l = -2n OR l = -n
∴ l = -2n
From (2), 3l + m + 5n = 0
∴ -6n + m + 5n = 0
∴ m = n
i.e. (-2n, n, n) = (-2, 1, 1)
∴ l = -n
∴ -3n + m + 5n = 0
∴ m = -2n
i.e. (-n, -2n, n) = (1, 2, -1)
(a1, b1, c1) = (-2, 1, 1) and (a2, b3, c3) = (1, 2, -1)
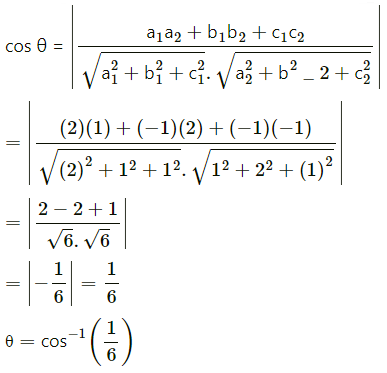

Question 31.
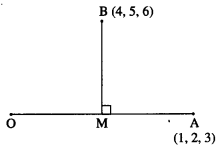
If Q is the foot of the perpendicular from P(2, 4, 3) on the line joining the points A(1, 2, 4) and B(3, 4, 5), find coordinates of Q.
Solution:
Let PQ be the perpendicular drawn from point P(2, 4, 3) to the line joining the points A(1, 2, 4) and B (3, 4, 5).
Let Q divides AB internally in the ratio λ : 1
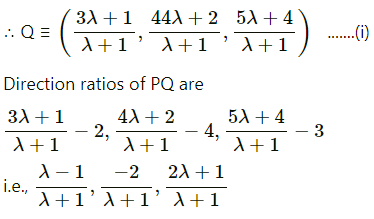
Now, direction ratios of AB are, 3 – 1, 4 – 2, 5 – 4 i.e., 2, 2, 1.

Coordinates of Q are,
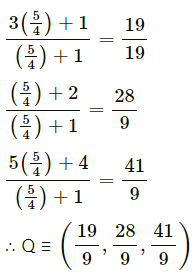
Question 32.
Show that the area of a triangle ABC, the position vectors of whose vertices are a, b and c is \(\frac{1}{2}[\vec{a} \times \vec{b}+\vec{b} \times \vec{c}+\vec{c} \times \vec{a}]\)
Question is modified.
Show that the area of a triangle ABC, the position vectors of whose vertices are \(\bar{a}\), \(\bar{b}\) and \(\bar{c}\) is \(\frac{1}{2}[\bar{a} \times \bar{b}+\bar{b} \times \bar{c}+\bar{c} \times \bar{a}]\).
Solution:
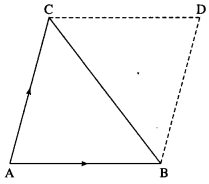
Consider the triangle ABC.
Complete the parallelogram ABDC.
Vector area of ∆ABC

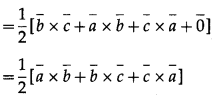
Question 33.
Find a unit vector perpendicular to the plane containing the point (a, 0, 0), (0, b, 0), and (0, 0, c). What is the area of the triangle with these vertices?
Solution:


Question 34.
State whether each expression is meaningful. If not, explain why ? If so, state whether it is a vector or a scalar.
(a) \(\bar{a} \cdot(\bar{b} \times \bar{c})\)
Solution:
This is the scalar product of two vectors. Therefore, this expression is meaningful and it is a scalar.
(b) \(\bar{a} \times(\bar{b} \cdot \bar{c})\)
Solution:
This expression is meaningless because \(\bar{a}\) is a vector, \(\bar{b} \cdot \bar{c}\) is a scalar and vector product of vector and scalar is not defined.
(c) \(\bar{a} \times(\bar{b} \times \bar{c})\)
Solution:
This is vector product of two vectors. Therefore, this expression is meaningful and it is a vector.
(d) \(\bar{a} \cdot(\bar{b} \cdot \bar{c})\)
Solution:
This is meaningless because \(\bar{a}\) is a vector, \(\bar{b} \cdot \bar{c}\) is a scalar and scalar product of vector and scalar is not defined.

(e) \((\bar{a} \cdot \bar{b}) \times(\bar{c} \cdot \bar{d})\)
Solution:
This is meaningless because \(\bar{a} \cdot \bar{b}, \bar{c} \cdot \bar{d}\) are scalars and cross product of two scalars is not defined.
(f) \((\bar{a} \times \bar{b}) \cdot(\bar{c} \times \bar{d})\)
Solution:
This is scalar product of two vectors. Therefore, this expression is meaningful and it is a scalar.
(g) \((\bar{a} \cdot \bar{b}) \cdot \bar{c}\)
Solution:
This is meaningless because \(\bar{c}\) is a vector, \(\bar{a} \cdot \bar{b}\) scalar and scalar product of vector and scalar is not defined.
(h) \((\bar{a} \cdot \bar{b}) \bar{c}\)
Solution:
This is a scalar multiplication of a vector. Therefore, this expression is meaningful and it is a vector.
(i) \((|\bar{a}|)(\bar{b} \cdot \bar{c})\)
Solution:
This is the product of two scalars. Therefore, this expression is meaningful and it is a scalar.
(j) \(\bar{a} \cdot(\bar{b}+\bar{c})\)
Solution:
This is the scalar product of two vectors. Therefore, this expression is meaningful and it is a scalar.
(k) \(\bar{a} \cdot \bar{b}+\bar{c}\)
Solution:
This is the sum of scalar and vector which is not defined. Therefore, this expression is meaningless.
(l) \(|\bar{a}| \cdot(\bar{b}+\bar{c})\)
Solution:
This is meaningless because \(\bar{a}\) is a vector, \(\overline{\mathrm{b}}+\overline{\mathrm{c}}\) is a scalar and the scalar product of vector and scalar is not defined.

Question 35.
Show that, for any vectors \(\bar{a}, \bar{b}, \bar{c}\)
\((\bar{a}+\bar{b}+\bar{c}) \times \bar{c}+(\bar{a}+\bar{b}+\bar{c}) \times \bar{b}+(\bar{b}+\bar{c}) \times \bar{a}=2 \bar{a} \times \bar{c}\)
Question is modified.
For any vectors \(\bar{a}, \bar{b}, \bar{c}\) show that
\(\begin{aligned}
&(\bar{a}+\bar{b}+\bar{c}) \times \bar{c}+(\bar{a}+\bar{b}+\bar{c}) \times \bar{b}+(\bar{b}-\bar{c}) \times \bar{a} \\
&=2 \bar{a} \times \bar{c} .
\end{aligned}\)
Solution:

Question 36.
Suppose that \(\bar{a}\) = 0.
(a) If \(\bar{a} \cdot \bar{b}=\bar{a} \cdot \bar{c}\) then is \(\bar{b}=\bar{c}\)?
Solution:
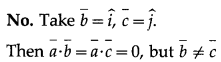
(b) If \(\bar{a} \times \bar{b}=\bar{a} \times \bar{c}\) then is \(\bar{b}=\bar{c}\)?
Solution:
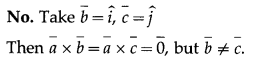
(c) If \(\bar{a} \cdot \bar{b}=\bar{a} \cdot \bar{c}\) and \(\overline{\mathrm{a}} \times \overline{\mathrm{b}}=\overline{\mathrm{a}} \times \overline{\mathrm{c}}\) then is \(\bar{b}=\bar{c}\)?
Solution:
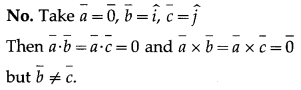
Question 37.
If A(3, 2, -1), B(-2, 2, -3), C(3, 5, -2), D(-2, 5, -4) then
(i) verify that the points are the vertices of a parallelogram and
Solution:

∴ opposite sides AB and DC of ABCD are parallel and equal.
∴ ABCD is a parallelogram.

(ii) find its area.
Solution:

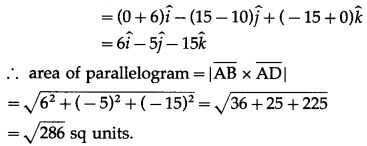
Question 38.
Let A, B, C, D be any four points in space. Prove that \(|\overline{A B} \times \overline{C D}+\overline{B C} \times \overline{A D}+\overline{C A} \times \overline{B D}|\) = 4 (area of ∆ABC)
Solution:
Let A, B, C, D have position vectors \(\bar{a}\), \(\bar{b}\), \(\bar{c}\), \(\bar{d}\) respectively.
Consider \(|\overline{A B} \times \overline{C D}+\overline{B C} \times \overline{A D}+\overline{C A} \times \overline{B D}|\)

Question 39.
Let \(\hat{a}, \hat{b}, \hat{c}\) be unit vectors such that \(\hat{a} \cdot \hat{b}=\hat{a} \cdot \hat{c}=0\) and the angle between \(\hat{b}\) and \(\hat{c}\) be\(\frac{\pi}{6}\).
Prove that \(\hat{a}=\pm 2(\hat{b} \times \hat{c})\)
Solution:
\(\hat{a} \cdot \hat{b}=\hat{a} \cdot \hat{c}=0\)
∴ \(\hat{a}\) is perpendicular to \(\hat{b}\) and \(\hat{c}\) both
∴ \(\hat{a}\) is parallel to \(\hat{b}\) × \(\hat{c}\)
∴ \(\hat{a}\) = m(\(\hat{b}\) × \(\hat{c}\)), m is a scalar.

Question 40.
Find the value of ‘a’ so that the volume of parallelopiped a formed by \(\hat{i}+\hat{j}+\hat{k}+a \hat{k}\) aand \(a j+\hat{k}\) becomes minimum.
Question is modified.
Find the value of ‘a’ so that the volume of parallelopiped formed by \(\hat{i}+a \hat{j}+\hat{k}, \hat{j}+a \hat{k}\) and \(a \hat{i}+\hat{k}\) becomes minimum.
Solution:
Let \(\bar{p}\) = \(\hat{i}+a \hat{j}+\hat{k}\), \(\bar{q}\) = \(\hat{j}+a \hat{k}\), \(\bar{r}\) = \(a \hat{i}+\hat{k}\)
Let V be the volume of the parallelopiped formed by \(\bar{p}, \bar{q}, \bar{r}\).
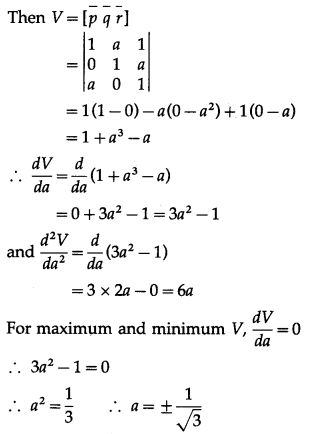
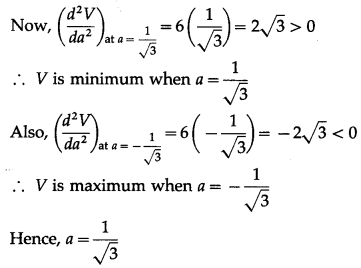

Question 41.
Find the volume of the parallelepiped spanned by the diagonals of the three faces of a cube of side a that meet at one vertex of the cube.
Solution:
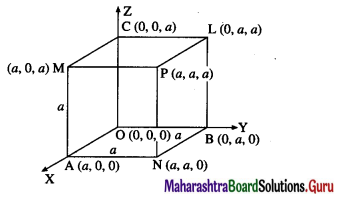
Take origin O as one vertex of the cube and OA, OB and OC as the positive directions of the X-axis, the Y-axis and the Z-axis respectively.
Here, the sides of the cube are
OA = OB = OC = a
∴ the coordinates of all the vertices of the cube will be
O = (0, 0, 0) A = (a, 0, 0)
B = (0, a, 0) C = (0, 0, a)
N = (a, a, 0) L = (0, a, a)
M = (a, 0, a) P = (a, a, a)
ON, OL, OM are the three diagonals which meet at the vertex O

Question 42.
If \(\bar{a}, \bar{b}, \bar{c}\) are three non-coplanar vectors, then show that \(\frac{\bar{a} \cdot(\bar{b} \times \bar{c})}{(\bar{c} \times \bar{a}) \cdot \bar{b}}+\frac{\bar{b} \cdot(\bar{a} \times \bar{c})}{(\bar{c} \times \bar{a}) \cdot \bar{b}}\) = 0
Solution:
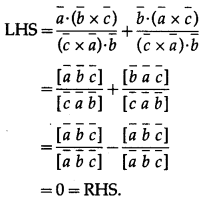
Question 43.
Prove that \((\bar{a} \times \bar{b}) \cdot(\bar{c} \times \bar{d})\left|\begin{array}{ll}
\bar{a} \cdot \bar{c} & \bar{b} \cdot \bar{c} \\
\bar{a} \cdot \bar{d} & \bar{b} \cdot \bar{d}
\end{array}\right|\)
Solution:
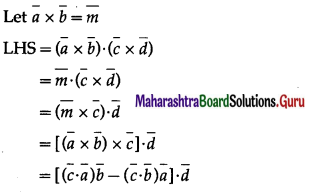

Question 44.
Find the volume of a parallelopiped whose coterminus edges are represented by the vector \(\hat{j}+\hat{k} \cdot \hat{i}+\hat{k}\) and \(\hat{i}+\hat{j}\). Also find volume of tetrahedron having these coterminous edges.
Solution:
Let \(\bar{a}\) = \(\hat{j}+\hat{k}\), \(\bar{b}\) = \(\hat{i}+\hat{k}\) and \(\bar{c}\) = \(\hat{i}+\hat{j}\) be the co-terminus edges of a parallelopiped.
Then volume of the parallelopiped = \([\bar{a} \bar{b} \bar{c}]\)
= \(\left|\begin{array}{lll}
0 & 1 & 1 \\
1 & 0 & 1 \\
1 & 1 & 0
\end{array}\right|\)
= 0(0 – 1) – 1(0 – 1) + 1(1 – 0)
= 0 + 1 + 1 = 2cu units.
Also, volume of tetrahedron = \(\frac{1}{6}[\bar{a} \bar{b} \bar{c}]\)
= \(\frac{1}{6}(2)=\frac{1}{3}\) cubic units.

Question 45.
Using properties of scalar triple product, prove that \(\left[\begin{array}{llll}
\bar{a}+\bar{b} & \bar{b}+\bar{c} & \bar{c}+\bar{a}
\end{array}\right]=2\left[\begin{array}{lll}
\bar{a} & \bar{b} & \bar{c}
\end{array}\right]\).
Solution:
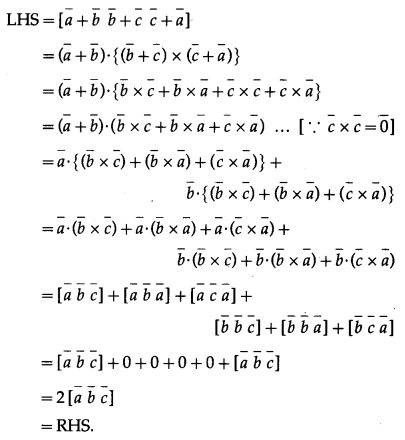
Question 46.
If four points A(\(\bar{a}\)), B(\(\bar{b}\)), C(\(\bar{c}\)) and D(\(\bar{d}\)) are coplanar then show that \(\left[\begin{array}{lll}
\bar{a} \bar{b} \bar{d}]+\left[\begin{array}{lll}
\bar{b} & \bar{c} & \bar{d}
\end{array}\right]+\left[\begin{array}{lll}
\bar{c} & \bar{a} & \bar{d}
\end{array}\right]=[\overline{\bar{a}} \bar{b} & \bar{c}
\end{array}\right]\)
Solution:

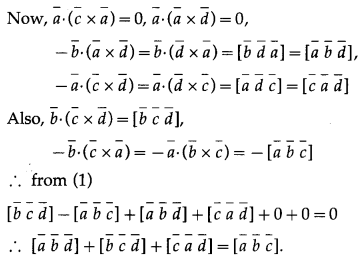
Question 47.
If \(\bar{a}\) \(\bar{b}\) and \(\bar{c}\) are three non coplanar vectors, then \((\bar{a}+\bar{b}+\bar{c}) \cdot[(\bar{a}+\bar{b}) \times(\bar{a}+\bar{c})]=-\left[\begin{array}{lll}
\bar{a} & \bar{b} & \bar{c}
\end{array}\right]\).
Solution:
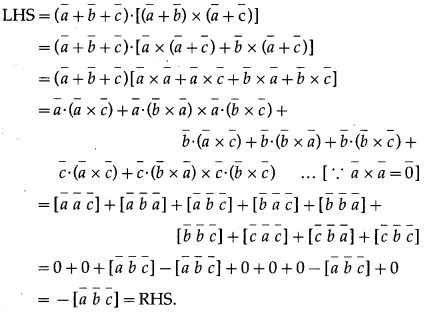

Question 48.
If in a tetrahedron, edges in each of the two pairs of opposite edges are perpendicular, then show that the edges in the third pair are also perpendicular.
Solution:
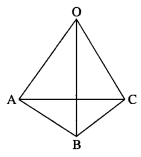
Let O-ABC be a tetrahedron. Then o
(OA, BC), (OB, CA) and (OC, AB) are the pair of opposite edges.
Take O as the origin of reference and let \(\bar{a}\) \(\bar{b}\) and 
∴ the third pair (OC, AB) is perpendicular.
Class 12 Maharashtra State Board Maths Solution

![]()
![]()
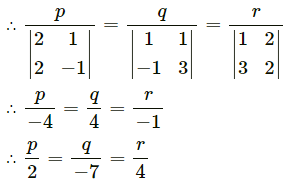
![]()
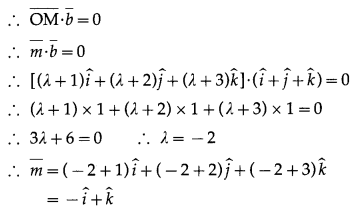


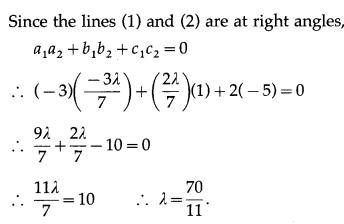

![]()
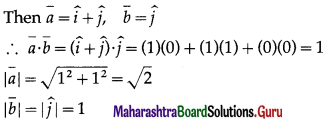
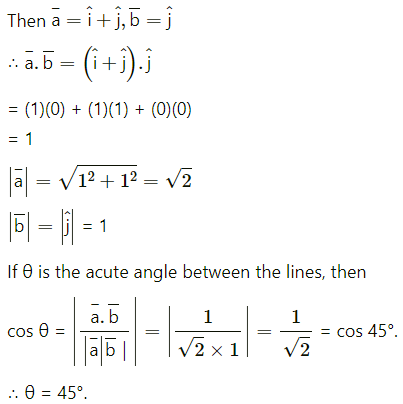
![]()
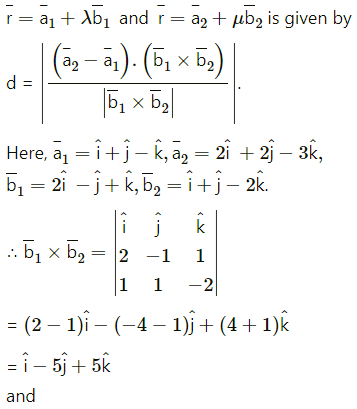
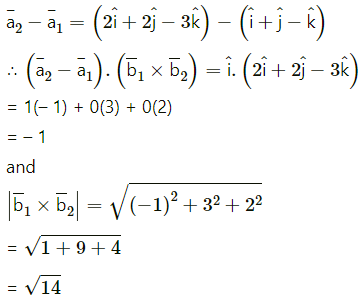
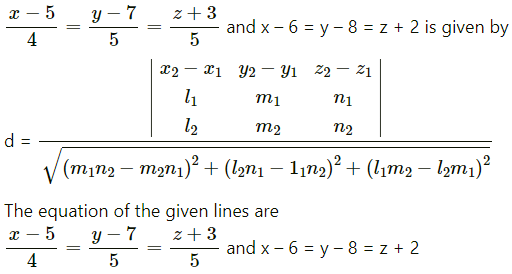
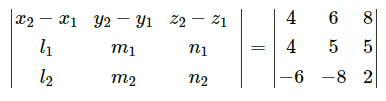
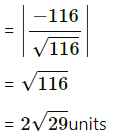

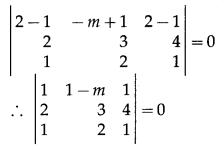
![]()
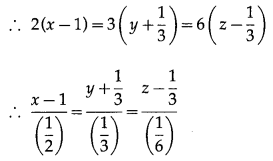




![]()